If All The Trees Fall in the Forest
DISCOVER, December 2002
Photography by Virginia Beahan & Laura McPhee One of thousands of coast live oaks killed by a forest plague in China Camp State Park. "The biggest lesson for me," a Forest Service pathologist says, "is that an incredible number of trees can die before most people take notice."  An advanced infection cracks the bark of a massive coast live oak, allowing sap to ooze out while opportunistic fungi attack. Phytophthora ramorum, the pathogen responsible for sudden oak death, is related to the organism that triggered the 19th-century Irish potato famine.  Young coast live oaks are the test subjects for a study at the University of California at Berkeley of treatments for sudden oak death. The stems are inoculated with P. ramorum. Then pathologists monitor the growth of the trees' lesions to determine the efficacy of a given treatment. Young coast live oaks are the test subjects for a study at the University of California at Berkeley of treatments for sudden oak death. The stems are inoculated with P. ramorum. Then pathologists monitor the growth of the trees' lesions to determine the efficacy of a given treatment.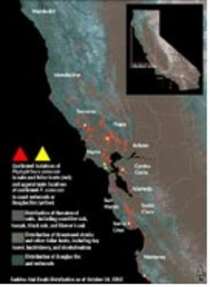 Sudden oak death has been found in more than two dozen species across nine plant families—from obscure shrubs to Douglas firs and redwoods. Douglas firs cover some 3.3 million acres, from California to British Columbia. Redwoods cover about 1.2 million acres, from Monterey to southern Oregon. Map courtesy of the Maggi Kelly Lab/Center for the Assessment and Monitoring of Forest and Environmental Resources (CAMFER) at the University of California at Berkeley; map designed by Karin Tuxen.  Forest pathologist Matteo Garbelotto is testing the effects of pruning wounds on trees infected with sudden oak death.  The quick and the dead are readily apparent in forests infected by sudden oak death. Forest pathologist David Rizzo was inspired to take up his career when he went hiking on the Shenandoah Mountain and came across the massive stumps of chestnut trees—vestiges of the last great forest plague. |
Two sleuthing scientists track down the cause of sudden oak death, a new disease that threatens every oak, redwood, and Douglas fir in the country.
One scorching June day two years ago, plant pathologist David Rizzo paid a visit to China Camp State Park. China Camp is a sanctuary of rolling golden hills and dense oak forest that hugs the shores of San Pablo Bay, California. It's a popular spot for hikers and campers, with a gentle wildness that makes it seem much more than 20 miles from San Francisco. But Rizzo was there on a serious mission, hot on the trail of a mysterious disease that was destroying the park's prized coast live oaks. Local rangers had spent years tearing out foreign interlopers like eucalyptus to make room for native trees. Now their work was unraveling. Month by month, the majestic, gnarled oaks were dying. "It was like losing old friends," ranger Patrick Robards says. "I kept saying: 'What's happening to my old friends?'"
Rizzo and three colleagues circled through one of the park's campgrounds, grateful for the shade cast by tall bay trees and broad oaks. There were plenty of ailing trees, marked by dark cankers, rivulets of sap, and trails of frass, telltale sawdust left by burrowing beetles. Rizzo pulled out his ax and chopped away a piece of the discolored bark. He could see a dark stain spreading underneath the healthy wood, its edges outlined by a crisp black line that marked the advancing infection. Rizzo slipped a sliver into a Baggie and moved on to the next tree.
The plague had been smoldering in the woods of Marin County for years. The first victims had been tanoaks, a distant relative of the stately trees we consider true oaks. Foresters called tanoaks a "trash species" and were almost happy to see them go. When county officials began investigating, the first experts on the case were entomologists. Because the dying trees were riddled with ambrosia and bark beetles—"there was a rain of sawdust coming down," one observer recalls—some entomologists assumed the trees were being killed by a beetle outbreak. The trees were sampled for pathogens, but the results came up negative, which only reinforced the beetle theory.
Then the problem began to spread. During the next few years, reports of dying trees came in from all over Marin County. "I had clients calling, saying, 'Oh my God, what's happening? I've lost three trees and more are bleeding,'" says Ken Bovero, an arborist in Mill Valley. "I've lived here all my life, and I was seeing something way out of context of anything I'd seen before." By the late 1990s, tens of thousands of trees were dead in four Bay Area counties, and their dry remains were a potential fire hazard.
Sudden oak death, as the catastrophe came to be called, is one of the most virulent forest epidemics ever to hit the United States. Its name is something of a misnomer: The disease is neither sudden nor limited to oaks nor invariably fatal. It now threatens forests in the East and South as well as California, and scientists confirmed in September that it has infected Douglas firs and redwoods—among our most valuable and beloved trees. Pathologists are investigating the disease, using both centuries-old techniques and DNA technology. They have conducted aerial surveys, worked on chemical cures, and found evidence of the disease as far away as Europe. In a modern world where any pathogen can hitch a ride across any ocean, their efforts may well write the book on how to cope with plant epidemics.
Still, on that June day in China Camp, Rizzo was dubious. Short and balding with an unassuming manner and a wry sense of humor, he isn't given to hysteria. He knew that oaks everywhere go through periodic die-offs, and he figured some familiar culprit was at work. So it came as a surprise when he walked into his lab at the University of California at Davis two days later and found his petri dishes teeming with tiny lemon-shaped spore sacs. It looked a bit like Phytophthora—a genus of funguslike organisms responsible for the Irish potato famine. But it wasn't like any phytophthora Rizzo had ever seen.
Nearly a century earlier, another exotic fungus made its way into America through trees imported from Japan. Known as chestnut blight, it was first discovered in the New York Zoological Garden in 1904 and was soon carried throughout the country by wind, rain, birds, and other animals. At the time, one in four trees from Maine to Mississippi was a chestnut; they were so plentiful that their creamy white blossoms seemed to cover the hills like snow in the spring. They were massive, too—often called "the sequoias of the East"—and to many, they were perfect: grand and fast-growing, with heartwood straight as a gun barrel that resisted warping and decay. The trees were used for everything from fine furniture to telephone poles. And every living thing in the chestnut belt, from hares to humans, relied on them. By the mid 1940s, virtually every mature chestnut in America had died—31/ 2 billion trees in all. The blight began by making the trees' bark fall away, then slowly killed them from the outside in. Today only chestnut sprouts survive in the forests, and the blight still kills them as soon as they reach the size of shrubs. James Hill Craddock, a biologist at the University of Tennessee at Chattanooga, calls it "without a doubt the worst ecological disaster to hit North America since the Ice Age."
Rizzo had no reason to believe the trees at China Camp were the leading edge of such a disaster. But when he saw the fungi in his lab, he immediately sent them to Matteo Garbelotto at the University of California at Berkeley for DNA analysis. Rizzo and Garbelotto had known each other slightly before being thrown together by the epidemic. Now, although their labs are 70 miles apart, they operate as a single entity, and their strengths are complementary. Rizzo excels at fieldwork honed in a variety of forests across the country. Garbelotto's extensive knowledge of molecular biology and genetic analysis has helped untangle the sudden oak death riddle.
When the samples arrived, Garbelotto had only recently joined the faculty at Berkeley and hadn't even had time to set up his lab. Still, he was very excited. He'd been arguing with entomologists for several months, disputing their beetle theory. Beetles infest dying trees, he said, but they don't cause the kind of lesions, or cankers, he'd seen on these trees. "It's like telling me Kaposi's sarcoma causes AIDS," he remembers thinking.
Working alone, on equipment donated by colleagues, Garbelotto began the long and tedious task of extracting DNA from the samples, amplifying the strands through a technique known as polymerase chain reaction, and then sequencing the hundreds of pairs of bases. Two months later, on a Friday night in August, he fed the finished sequence into an online data bank to compare with other known gene sequences.
When he got the results, he called Rizzo at home immediately. "It's something completely new," he crowed—a species of phytophthora never seen before. He made Rizzo try to guess the closest genetic match that he could find. When Rizzo drew a blank, Garbelotto told him it was Phytophthora lateralis, an exotic pathogen of unknown origin that had been destroying Port Orford cedars in Oregon and northern California for some 50 years.
The relationship between the two pathogens was unclear, but the similarities suggested possibilities. The two microbes might have emigrated from the same place, or P. lateralis might have mutated or mated with a different phytophthora species to form the new organism. Perhaps changes in the forest environment—fire suppression, for example—had produced trees that were susceptible to an otherwise benign microbe.
Before those possibilities could be sorted, Garbelotto received a visit from Clive Brasier, a British pathologist and the world's expert on phytophthoras in trees. Brasier agreed that the China Camp fungus looked like something new. But a few weeks later, he called to tell Garbelotto of another new and strikingly similar microbe, this one found on diseased rhododendrons in Germany and the Netherlands. The Europeans had dubbed it Phytophthora ramorum.
In the past, it would have taken months or years to swap and compare cultures. Now, with the click of a mouse, scientists on either side of the Atlantic exchanged computer files containing the organisms' gene sequences. They were astonished to find that the DNA fingerprints looked much the same.
"That discovery changed everything," Rizzo recalls. A disease afflicting forest oaks is bad enough, but at least oaks don't move. Rhododendrons—well, rhododendrons are shipped all over the world. In fact, a quick check of a local nursery showed that their rhododendrons were already harboring the pathogen. They didn't develop cankers or bleeding trunks, and they didn't all die—their main symptoms were black spots on leaves and small branches. But that was even more troubling in some ways: The organism moved through the air, apparently, and it produced different infections depending on the host. "That," Garbelotto says, "was something we'd never seen before in temperate forests."
Garbelotto, 36, cuts a dashing figure in the flannel-and-fleece world of forestry. Voluble, excitable, always on the go, he is both charming and slightly arrogant. He favors black leather jackets and stylish sunglasses, sports a hip chip of beard under his lip, and speaks with a burbling Italian accent. He was born to an aristocratic Venetian family and was raised in the Italian Alps, where he foraged for herbs and edible plants with his grandmother. "I've always had a passion for forests," he says, but traditional forestry didn't interest him. "I wanted something hands-on, where I could cook a bit." The answer was pathology, using molecular biology to diagnose tree diseases.
One of the central principles of plant pathology is Koch's postulate, Garbelotto said one afternoon, sitting in his office at Berkeley. It works this way: If you want to prove that a certain pathogen causes a disease, you first have to detect it in a host at every stage of that disease. You then have to isolate it, grow it in a culture, infect another host with it, and see if the same symptoms develop. If they do, you have to isolate the pathogen one more time and make sure it's identical to the one you isolated before. This procedure has proved nearly foolproof since German bacteriologist Robert Koch first formulated it in the late 19th century. But it can be excruciatingly slow work. In the case of sudden oak death, Rizzo says, the pathogen was "a fiend to isolate." Just developing the right extraction technique for any given tree could take months.
California didn't have time for that. Sudden oak death was showing up everywhere. Aerial and ground surveys turned up infected trees—including members of another species, the black oak—across a more than 200-mile stretch along the coast from Napa Valley to Big Sur. So Rizzo and Garbelotto needed a shortcut. They didn't abandon Koch's postulate—indeed, one of their first acts was to set up plots in Marin for inoculation experiments. However, they also decided to use DNA analyses to track the microbe's path. Rizzo made treks into the field, grabbing any spotty leaves he saw, and sent them to Garbelotto, who had developed a test for P. ramorum. If the DNA analysis identified P. ramorum in the sample, they would inoculate a healthy specimen of the latest victim with a ready-made culture of P. ramorum. This allowed them to test Koch's postulate without having to painstakingly extract the microbe from a sick plant. "This is going to be the approach of the future," Garbelotto says.
By June 2002, Garbelotto's DNA analysis had found phytophthora on 30 hosts across nine plant families, many of them unrelated. "It's as if I were to tell you that the same disease you can get, your cat, your dog, and your bird can get—even the kangaroos in the zoo can get," Garbelotto says. Certain that there must be some mistake, Rizzo decided to send his colleague a ringer—a ratty-looking bay laurel leaf with no relation to any host then known. Garbelotto phoned the next day: "Do you know that bay leaf had more phytophthora DNA than anything else you ever sent me?"
At first, few outside the Bay Area paid much attention to the growing list. Then about a year ago, Rizzo and Garbelotto detected the pathogen on the dying sprouts of redwood trees—the beloved symbol of California and one of its most valuable timber species. Rizzo spent three days fielding calls from The New York Times, the BBC, and other media. By September, he was back on the front pages with more grim news. Tests confirmed redwoods were a host and had turned up yet another victim: Douglas firs, the dominant tree of the Pacific Northwest. Together the two species account for about half the wood harvested in California each year, worth $3 billion. Governor Gray Davis fired off a letter to President Bush demanding $10 million to combat the "grave risks" posed by the disease.
Yet it wasn't—and still isn't—clear what the findings mean. "The bottom line?" says Rizzo. "We don't know." The diseased Douglas firs were found in only one site, where they were surrounded by infected bay trees. In the lab, firs react strongly to the pathogen, but in the field, the main effect is the wilting of new foliage in saplings. While P. ramorum is widespread among redwoods, only the sprouts seem to be succumbing, and most of those die anyway on the fiercely competitive forest floor. The most pressing question of all remains unanswered: Can the pathogen infect, much less fell, a mature forest conifer?
Early experiments have had mixed results. When researchers put the fungus under the bark of living redwoods, the trees developed lesions, then recovered. But studies have also found that red oaks and pin oaks, two species found all over the East Coast, are as vulnerable under artificial conditions as tanoaks, which invariably succumb. Rizzo and Garbelotto are trying to determine whether the pathogen can be spread through bark mulch sold for gardening.Whether it could take hold in eastern oaks under natural conditions isn't clear. But if it does, "it could well exceed anything we have seen," says Everett Hansen, a forest pathologist at Oregon State University.
Rizzo, Garbelotto, Hansen, and other researchers have now assembled a plausible scenario of how the fungus spreads. All phytophthora release tiny football-shaped sacs containing spores that travel through water. But while most phytophthora infect from the roots up, P. ramorum seems to work from the top down. "The spores swim out on the water film of leaves and settle down to germinate," Hansen says, "sending hyphae, little filaments, into the leaf or stem, which then grow inside the host tissue and extract sugars from the leaf or bark." That explains the leaf spots that appear on so many non-oak hosts. Each blackened leaf tip indicates the presence of roughly 10,000 spores. In most hosts other than oaks, P. ramorum only shrivels leaves and kills off twigs and small branches.
Non-oak hosts may be the driving force of the epidemic. They pass on P. ramorum to susceptible oak trees, just as rats and fleas can transmit plague bacteria. Since such hosts are far less likely to die than oaks, the microbe can persist in a forest long after the oaks are gone. This explains why the hardest-hit trees are not in pure stands of oaks but in woods where oaks are surrounded by bay laurel, madrona, rhododendron, or other species. One study found coast live oaks were 10 times more likely to be infected when bay laurel was nearby.
That theory also explains the epidemic's timing. Reports of dying trees shot up in the spring of 1996 and 1999, both of which came a year after the extraordinarily wet winters caused by El Niño. The wind and rain served as autobahns for the spores. "They're calling for another El Niño in 2003," Rizzo says. "I get a little nervous because if that's the case, be prepared for things to be ugly in 2004 or 2005 and in new places." The epidemic has already jumped from the Bay Area to one of California's northernmost counties and to a forest in southern Oregon.
Outside California, the most likely spots for P. ramorum to show up are the Appalachian states. Those areas have the same cool, moist climate as northern California, they boast lots of rhododendron nurseries, and their forests have a similar mix of oaks and vulnerable understory trees. The Forest Service is planning aerial and ground surveys in those forests as well as in forests around rhododendron nurseries. But one pathologist admits the search is like looking "for a needle in a haystack."
Meanwhile, Garbelotto and Rizzo have been working on ways to slow down the epidemic. Experiments have already pointed to phosphonates as a cure. These fertilizers appear to shrink lesions in coast live oaks. Even if the treatment works, there's no way to administer it to an entire forest. Then again, Garbelotto says, "maybe all we need to save are three or four oaks per acre, and that will provide enough genetic material to maintain the local population." In Australia, where an outbreak of another type of phytophthora has affected 2,000 different forest species, such an approach has saved several species from extinction.
Finally, but perhaps most important, Garbelotto has been working to develop a DNA assay that will identify trees that are resistant to P. ramorum. The acorns from those trees can then be planted to regenerate the forests. Elm lovers have used the same techniques to develop a resistant line of Dutch elms, and a campaign to plant a million of the trees is under way. "Stopping the epidemic is impossible," Garbelotto says, yet "if things work, we may be able to prevent even larger numbers of trees from dying. We may be able to make sure that what happened in Marin County doesn't happen again."
Sudden oak death is invariably described as devastating, but its effects aren't always obvious to an untrained eye. When arborist Ken Bovero takes visitors on a tour of a particularly hard-hit site—a gorgeous canyon running through central Mill Valley in Marin County—he sees nothing but desolation: both tanoaks and coast live oaks marked by rivulets of wine-smelling sap, beetle boreholes, and warty black outgrowths of opportunistic fungi. But where Bovero sees a ridge scattered with skeletons of fallen trees, others may see only a cool and verdant refuge from the surrounding thrum of urban life.
Which raises the question: What should a forest look like? Some contrarians say concerns about exotic pathogens are overblown, that it's no use devoting resources to stanch an epidemic. Diseases—invasive and native—help prevent overcrowding and maintain a healthy equilibrium in forests, says John Castello, a forest pathologist at the State University of New York at Syracuse. "It's like culling a herd." Forests are forever changing, he says. Sure, the American chestnut is gone, "but oaks and hickories have taken its place. If you're a chestnut connoisseur, you don't want to hear this. But the forest doesn't give a damn; it's adjusting."
Forest plagues are not new; fossil records document destructive outbreaks tens of thousands of years ago. Yet natural forces didn't introduce diseases like sudden oak death or chestnut blight—humans did. And as humans trade and travel across the globe, the trickle of pathogens hopping oceans and jumping borders has become a flood. Federal and state inspectors now intercept about 53,000 pathogens, insects, and noxious plants each year, according to a National Academy of Sciences report, and they examine only about 2 percent of all the arriving cargo and baggage. Only a tiny fraction of those that slip past take hold, but they tend to be harmful. Exotic pathogens are now decimating butternut trees in the Northeast, flowering dogwoods in the South, and Port Orford cedars in the Pacific Northwest, among others. Equally damaging are émigré insects, such as gypsy moths, glasswing sharpshooters, and Asian longhorn beetles. It's easy to see why many ecologists consider invasive species a major environmental threat.
A century ago, chestnut trees were the backbone of the Appalachian food chain. When they disappeared, some experts believe, they took the passenger pigeon with them, as well as many wild turkeys and other game species. In California, researchers are just starting to study how sudden oak death could affect wildlife. Oak habitat alone covers 11 million acres in California, providing food and shelter for more than 200 species of mammals, from black bears, mule deer, and coyotes to jackrabbits, chipmunks, and deer mice. More than 100 species of birds nest in oaks, including the meadow woodpecker, the mourning dove, and the band-tailed pigeon. Add in other susceptible plant species, like manzanita, bay laurel, huckleberry, and madrona, and virtually every type of animal that lives in California's mixed evergreen forests could be affected.
"There's an apocalyptic vision I have, and it's not too far-fetched," says Craig Lorimer, a professor of forest ecology and management at the University of Wisconsin at Madison. "Because diseases and insects spread slowly, people don't notice the change. Relatively few people remember what the chestnut forests looked like, so there's a whole new generation who don't know they're missing anything. When you have tree species after tree species disappearing, what worries me is there will still be trees and shrubs, and people will think their forest is beautiful. But to those who know what the forest once looked like, it will look like the ruins of an ancient civilization."